Most reactive halide towards S_(N^(1)) reactions is Tertiary butyl chloride is most reactive in SN1 reaction. Which halogen is more reactive in SN1 reaction? Is Cl or Br more reactive?Īlthough the bromine nucleus is more positively charged than the chlorine nucleus, the increase in the radius and the extra shielding in the bromine atom outweigh this factor, which means that an electron is more easily attracted into the outer shell of a chlorine atom than that of a bromine atom, so chlorine is more … Halogens are more reactive than hydrogen because, in case of halogens they have 7 valence electrons in their valence shell so they acquire 1 more electron to complete their octet attaining noble / inert gas configuration. Which is more reactive halogen or hydrogen? Its chemical activity can be attributed to its extreme ability to attract electrons (it is the most electronegative element) and to the small size of its atoms. The reaction is faster.įluorine (F), most reactive chemical element and the lightest member of the halogen elements, or Group 17 (Group VIIa) of the periodic table. Has to be warmed and the iron wool heated. Reacts with heated iron wool very quickly. 
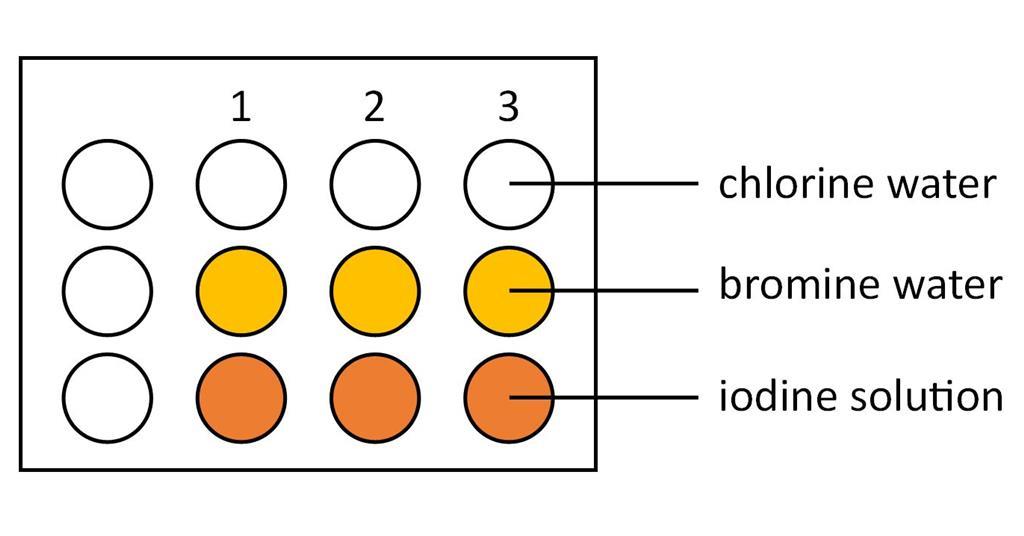
Which is the third most reactive halogen?įluorine is the most reactive element of all in Group 7….Reactivity of halogens. The order of reactivity is chlorine > bromine > iodine. 
Use the results in the table to deduce an order of reactivity, starting with the most reactive halogen. Fluorine is one of the most reactive elements. Halogens can gain an electron by reacting with atoms of other elements. This reactivity is due to high electronegativity and high effective nuclear charge. Halogens are highly reactive, and they can be harmful or lethal to biological organisms in sufficient quantities. What is the order of reactivity of halogens? 4 Which halogen is most likely to react?.2 Which is the third most reactive halogen?.1 What is the order of reactivity of halogens?.The stability of oxides increases with the increase in number of oxygens in the compound. Iodine oxides are highly stable because of greater polarizability of bond between iodine and oxygen. The kinetic and thermodynamic studies show that stability of oxides of halogen follows the order : I > CI > Br. `I_2O_5` is a very good oxidising agent and is used in the estimation of carbon monoxide. The iodine oxides, `I_(2)O_(4), I_(2)O_(5), I_(2)O_(7)` are insoluble solids and decompose on heating. (ii) Very less polarizability of Br-0 bond. The bromine oxides are least stable because : (i) Bromine cannot form multiple bonds with oxygen. 
The bromine oxides, `Br_2O, BrO_(2), BrO_3` are the least stable halogen oxides (middle row anomanously) and exist only at low temperatures. The stability of oxides of chlorine can be explained due to fact that multiple bond formation between oxygen and chlorine takes place due to availability of d-orbitals in chlorine. They tend to explode.`ClO_2` is used as a bleaching agent for paper pulp and textiles and in water treatment. Chlorine oxides, `Cl2_O, ClO_2, Cl_2O_6` and `Cl_2O_7` are highly reactive oxidising agents. `O_2F_2` oxidises Pu to `PuF_6` and the reaction is used in removing plutonium as `PuF_6` from spent nuclear fuel. Fluorides of oxygen are `OF_2` and `O_2F_2`. The compounds of oxygen with fluorine are called fluorides because fluorine is more electronegative than oxygen. Solution : Halogens form many oxides with oxygen but most of them are unstable. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
